
Studies and Medical reports PeniMaster PRO
PeniMaster PRO for post-operative treatment of prostate cancer Clinical study 2015/2016
- Prostate cancer: widespread tumour among men of middle age and older.
- Number of diseases risen by more than 120% within 10 years.
- Removal of the prostate (prostatectomy) is an established and life-prolonging process in the event of prostate cancer.
- Frequent and significant side effects:
- Libido (sexual longing) and erectile capacity suffer under the removal of the prostate.
- The length of the penis decreases (the penis becomes shorter).
- Ejaculation problems.
- Objective of the study: to study the possibilities for improving quality of life after prostatectomy.
- Participants in the clinical study: 60 men between 60 and 69 years of age, after cancer-induced removal of the prostate.
- Clinical study according to international ethical clinical standards.
- Erectile dysfunction in the case of prostate cancer was also investigated as part of the study.
- There were three comparative groups, each comprising 20 men:
- Group 1: received no preventative treatment for the preservation of the length of the penis and of erectile function.
- Group 2: received PDE5 inhibitors for three months after the operation
- Group 3: additionally used the penis expander PeniMaster PRO for at least three hours every day.
POSSIBILITIES OF COMBINATION TREATMENT FOR PRESERVING THE SEXUAL FUNCTION IN PATIENTS AFTER RADICAL PROSTATECTOMY FOR LOCALIZED PROSTATE CANCER (1)
Prostate cancer (PC) is one of the most common malignant tumors in the middle-aged and elderly male population, occupying the 4th place in terms of cancer morbidity in Russia [1]. In the past decade, the PC incidence rate has increased by more than 120.5% [2].
Surgery is known to be the main and most widely used method for treating localized prostate cancer. Surgical treatment provides onco-specific survival compared to watchful waiting [3].
According to the clinical guidelines of the European Association of Urology, radical prostatectomy (RPE) is a standard therapy for localized prostate cancer if the life expectancy of a patient exceeds 10 and more years [4, 5].
The problem of increasing the life expectancy of patients after RPE is inseparably associated with the problem of improving the quality of life. It has been established that RPE entails a number of complications that affect the quality of life, such as lowered libido, erectile dysfunction, shortening and bending of the penis, ejaculation problems [6, 7].
Sexual function recovery after RPE has been studied for a long time. However, despite the published findings, neither national nor foreign authors have reached a unanimous opinion concerning the state of erectile function after RPE [8, 9, 10, 11, 12, 13]. A significant influence of erectile function on quality of life causes the necessity and urgency of further investigation of the erectile function state in patients who have undergone RPE [14, 15, 16, 17].
RESEARCH GOAL: evaluating the possibility of combination treatment for sexual function in patients after radical prostatectomy to treat localized prostate cancer as a factor in increasing quality of life.

“Municipal Clinical Hospital No. 3 M. A. Podgorbunsky”, Kemerovo, ФГБУ ГНЦ ФМБЦ им. А.И. Бурназяна ФМБА России [(Federal State Budgetary Institution State Research Center Federal Medical Biophysical Center) A. I. Burnazyan FMBA (Federal Medical-Biological Agency) of Russia], Moscow2
Kyzlasov Pavel PhD Head of the department urology
Federal Medical Biological Agency Burnasyan Federal Medical biophisical Centre
www.fmbcfmba.ru
Pomeshkin Evgeny PhD Head of the department urology, Chief urologist Kemerovo
M.A. Podgorbunsky City Municipal Hospital
www.gkb3.ru
MATERIALS AND METHODS:
The present research includes 60 men of the average age of 64.6±4,7 years with verified localized prostate cancer, who underwent treatment in the urology department of ФГБУ ГНЦ ФМБЦ им. А.И. Бурназяна ФМБА России , Moscow.
All research studies with the participation of patients complied with the ethical standards of the Bioethical Committee developed in accordance with the World Medical Association's Declaration of Helsinki “Ethical Principles for Medical Research Involving Human Subjects”. Patients signed a voluntary informed consent to participate in the research study.
The research entry criteria were men not older than 70 years old having no heavy somatic pathology or diabetes and being interested in preservation of sexual function after the surgery.
In the course of the study, the International Index of Erectile Function of all the patients was evaluated in accordance with IIEF-5, and the patients’ quality of life was assessed in accordance with the QOL (Quality of Life) questionnaire. Also, the length of non-erect penis (L) of all the patients was measured three times and averaged, and the penile rigidity was evaluated according to the penile erection hardness scale (EHS) (table 1).
Table 1. The penile erection hardness scale (EHS)
| Degree | |||
| 1 | 2 | 3 | 4 |
| The penis is increased in size but not hard enough. | The penis is hard but not hard enough for penetration. | The penis is hard enough for penetration but still not completely hard. | The penis is completely hard and fully rigid. |
Control measurements were carried out before the surgery, on the 7th, 30th and 90th day after the surgical intervention.
Initially, none of the patients participating in the research study had any significant differences in clinico-anamnestic features. According to the research design, patients were arbitrarily divided into three groups using a random number table. The patients in the first group (control group, n=20) did not receive any specific preventive medication for preserving penile length and erectile function. The second group of patients (n=20) took PDE-5 inhibitors daily in a dose of 5 mg during 3 months in the postoperative period; the third group of patients (n=20) combined the intake of PDE-5 inhibitor in a dose of 5 mg with the use of a vacuum extender for three months. The vacuum apparatus PeniMaster PRO was used as an extender in the research study, the patients of the third group carried the extender daily for not less than three hours a day during three months.
The obtained data were statistically processed using the STATISTICA 6.0 software. The normal distribution hypothesis was checked with the aid of the Shapiro-Wilk test. An average value (M) and a standard deviation (SD) are given for each of the continuous quantities exhibiting normal distribution.
Values of p<0.05 were statistically significant. The three groups were compared using the N-criterion of the Kruskal-Wallis test; at p<0.05, a paired comparison of groups was used employing the Mann-Whitney test with the application of the Bonferroni adjustment. The dynamics parameters were evaluated using the Wilcoxon criteria.
PeniMaster PRO for post-operative treatment of prostate cancer Clinical study 2015/2016
- Results:
- Group 1: shortening of the penis after 3 months: approx. - 1.6 cm / - 12.45%
- Group 2 (with medication):
Shortening of the penis after 3 months: approx. - 1.2 cm / - 10.26% - Group 3 (with PeniMaster PRO penis expander):
ENLARGEMENT of the penis after 3 months: approx. + 0.75 cm / + 5.5% - Difference in penis length group 1 (untreated) / group 3 (with PeniMaster PRO): approx. 18%
FINDINGS AND DISCUSSIONS
The analysis of the penile length measurement results proved that, 7 days after the surgery, the average shortening of the penis in the control group measured 0.73±0.08 cm, 30 days after the surgery it was 0.95±0.06 cm, and 3 months later it was 1.635±0.12 cm compared to the measurements made before the surgery (Fig. 1). Thus, 3 months after the surgery, the penile length shortening amounted to 12.45%.
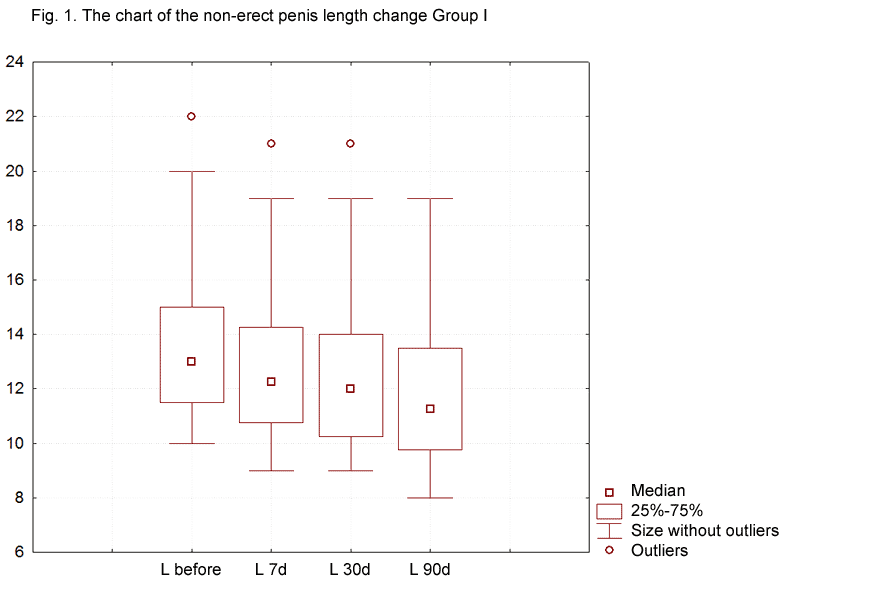
In the second group of patients who took PDE-5 inhibitors, the average shortening of the penile length measured 0.75±0.08 cm 7 days post-surgery, 1.125±0.07 cm 30 days later and 1.175±0.08 cm 3 months later compared to the measurements made before the surgery (Fig. 2). According to the obtained data, the penile length shortening in the second group amounted to 10.26%, which is 2.19% less than in the control group.
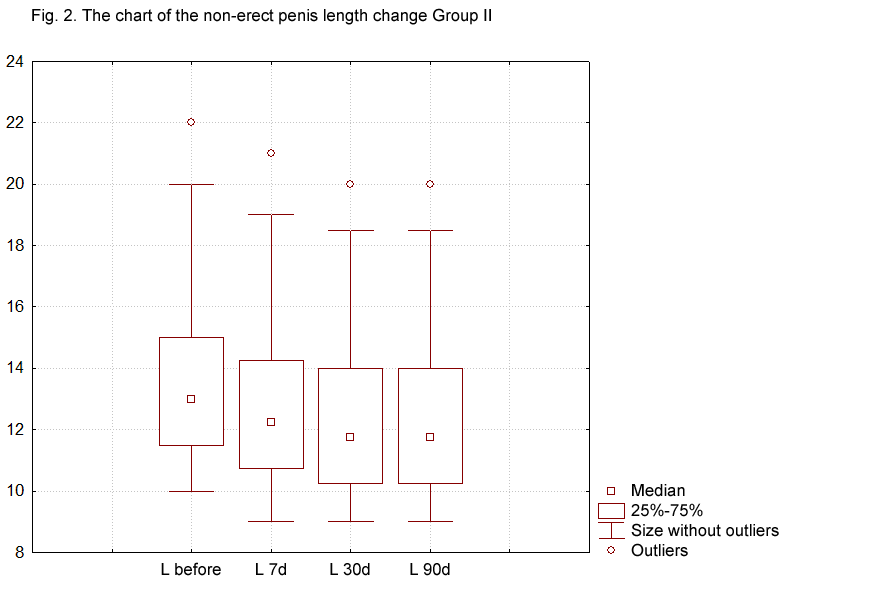
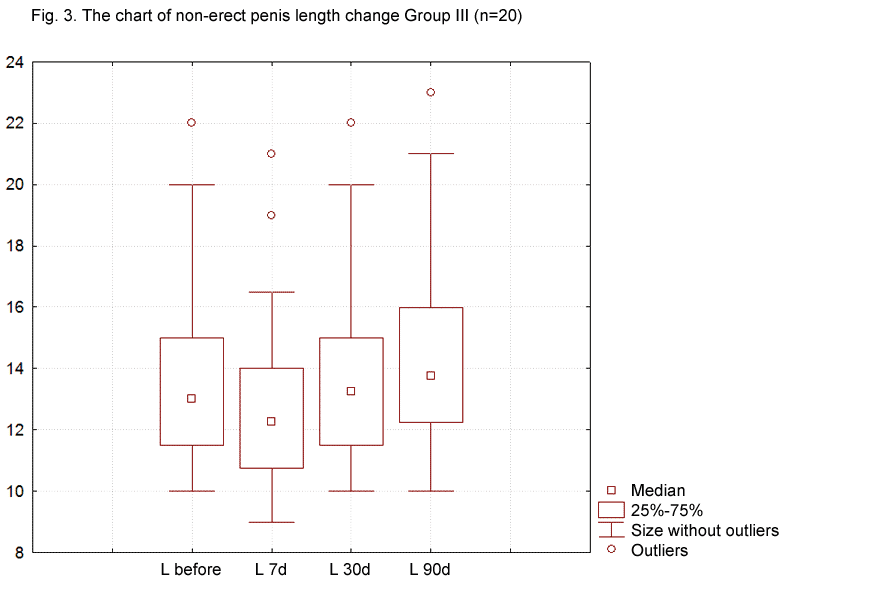
The third group of patients, who combined the intake of PDE-5 inhibitors with the use of a vacuum extender, saw the average shortening of the penis measure 0.78±0.1 cm 7 days post-surgery (Fig. 3). After 30 days, a statistically significant increase in penis length by 0.175±0.08 cm was observed and after 3 months, the length increase measured 0.75±0.08 cm. Thus, despite the penis shortening by 5.7% by the 7th day, an increase in the penis length by 5.5% was observed by the 90th day compared to the initial data.
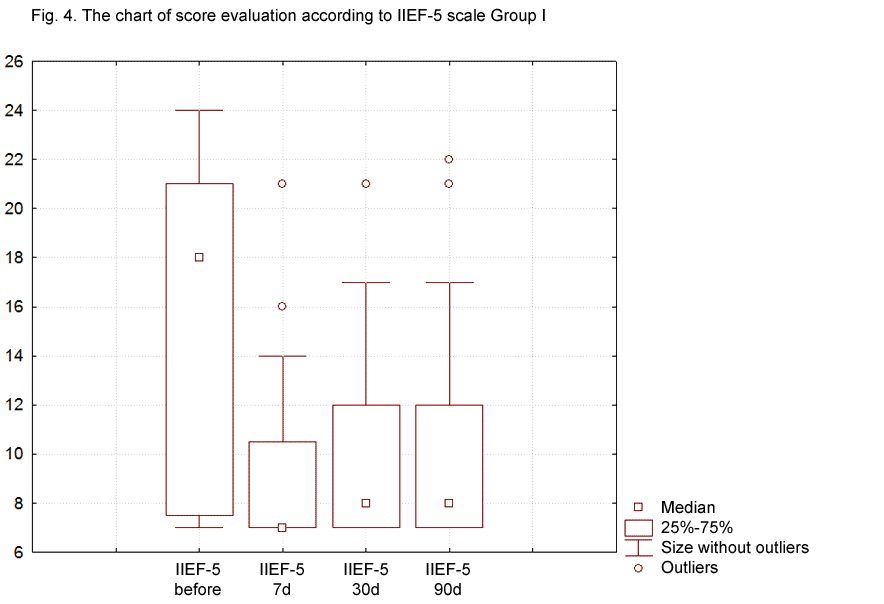
While assessing the test results in accordance with IIEF-5 Questionnaire, the following data were obtained: in the first group, the total score of the erectile dysfunction intensity before the surgery was equal to 16.5 points, by the seventh day after the surgery, it reduced by 6.45±1.3 points, which resulted in 9.7 points on average (Fig. 4). By the 30th and 90th days, an insignificant increase to 10.1 and 10.2 points, respectively, was registered.
In the second group, the total score of the erectile dysfunction intensity decreased by 6.45±1.3 points by the 7th day after the surgery, which resulted in 9.7 points (Fig. 5). By the 30th and 90th days, an increase in the total score was registered, namely 13.2 and 13.4 points respectively. When assessing the total score of erectile dysfunction in the third group, the obtained data did not exhibit any statistically significant difference from the results of the second group (Fig. 6).
PeniMaster PRO for post-operative treatment of prostate cancer Clinical study 2015/2016
- After 90 days, the penis rigidity of the non-treated test participants from group 1 was no longer sufficient for the practice of sexual intercourse.
- In groups 2 and 3 (with PeniMaster PRO), the test participants achieved an erection rigid enough for sexual intercourse.
- Other studies also show a shortening of the penis after prostate removal by 1-3 cm within one year.
- The study shows that PeniMaster PRO not only preserves the length of the penis after removal of the prostate, but improves it.
- Not only recovery from the disease, but also rehabilitation from after-effects is important in modern medicine.
- There are no standard specifications for treatment after prostatectomy.
FINDINGS AND DISCUSSIONS
When evaluating penis rigidity according to PHS, the following results were obtained: in the first group, the degree of erectile rigidity was 3.1 before the surgery, by the 7th day after surgery, it reduced by 1.22±0.2. By the 30th and 90th days the result was 2, which corresponds to a sufficiently hard penis, but not hard enough for penetration and sexual intercourse.
The data obtained in the second and third groups did not differ and were characterized by a decrease in penis hardness during the first 7 days, and then by an increasing tendency up to the 3rd degree, which is characterized by a hardness sufficient for penetration.
In the first group, the quality of life according to the QOL scale was 2.1 points before the surgery; 7 days after the surgery it reduced by 2.05±0.3 points, after which a positive increasing tendency was observed; thus, the average life quality level measured 3.7 points on the 30th and 90th days after the surgery.
In the second and third groups, the lowering of the quality of life to 4.15 points was also registered by the 7th day, and by the 30th and 90th day, the quality of life increased to 2.4 points in the second group and to 2.6 points in the third group. No statistically significant differences in the life quality level were observed between the second and third groups.
According to the literature data, no convincing data is currently available on the application of PDE-5 inhibitors for the rehabilitation of sexual function in males after radical prostatectomy for localized prostate cancer. Also, there is no unanimous opinion concerning doses, terms and time of taking PDE-5 inhibitors by this category of patients.
According to the findings by A. Briganti and co-authors [18], in the group where patients constantly took PDE-5 inhibitors, erectile dysfunction was observed much less frequently (37%) compared to the control group (73%, p<0.001), which is generally comparable to the data obtained in the course of the research study. After 3 months, the erectile dysfunction incidence was 75% (15 patients) in the control group, less by 15% (which corresponds to 60%) in the second group (12 patients) and less by 20% in the third group, namely 55% (11 patients).
Some research studies showed evidence of penis shortening by1-3 cm in patients after radical prostatectomy during a one-year observation period. [19, 20] This research study considered changes in penis length during a period of three months after surgical treatment, which demonstrated the length reduction by up to 1.64±0.12 cm in the control group, the penis shortening degree in the second group was lower: 1.175±0.01 cm on the average.
Possible mechanisms responsible for penile length shortening may be cavernosal fibrosis, caused by apoptosis of the smooth muscle cells of cavernous bodies after injury to the pelvic splanchnic nerves (nervi erigentes).
Earlier studies [21] showed that using vacuum extenders allows the penis length to be preserved after radical prostatectomy. At the same time, opinions differ as to using vacuum extenders considering the cavernosal fibrosis hazard resulting from cavernous body ischemia, acidosis and insufficient relaxation of smooth muscles [22]. Our findings show that combining daily intake of PDE-5 inhibitors with daily use of a vacuum extender makes it possible not only to preserve the penis length, but also to increase the penis length by 0.75±0.01 cm.
In modern society, quality of life is an essential part of a patient’s general health and wellness. Numerous research studies dedicated to life quality assessment proved the importance and necessity of developing and introducing patient rehabilitation programs. At present, no distinct and recommended rehabilitation algorithms with proven efficiency are available for patients after radical prostatectomy [7, 14, 15, 16, 17].
PeniMaster PRO for post-operative treatment of prostate cancer Clinical study 2015/2016
- The combination of PDE5 inhibitors and PeniMaster PRO prevents penis shrinkage and enlarges the penis after prostate removal.
- When untreated, erectile dysfunction occurs up to 20% more frequently after prostate removal.
- Erectile dysfunction has a significant, negative impact on quality of life.
CONCLUSION
The findings of the research study allow for the formulation of the following conclusions:
- No uniform, standard approach is currently available regarding sexual function recovery in patients after radical prostatectomy for localized prostate cancer
- A combination of daily intake of PDE-5 inhibitors and the use of a vacuum extender makes it possible not only to prevent penis shortening, but also to increase its length by 0.75±0.01 cm;
- The rate of incidence of erectile dysfunction after radical prostatectomy was observed to be lower by 15 and 20% in the patients of groups I and II respectively compared to the control group;
- Erectile function significantly lowers the quality of life of postoperative patients.